Research Topics of the Engelhard Group

Research area:
Research in our group revolves around analytical chemistry with a special focus on ultra trace elemental and molecular mass spectrometry, novel methods for nanoanalysis and nanomaterials characterization, direct surface analysis with ambient desorption/ionization mass spectrometry, fundamentals, and instrumentation. We use state-of-the-art instrumentation including ICP-MS, ICP-OES, HPLC-ESI-HRMS, GC-EI-MS, FAPA-HRMS and TOF-SIMS.
Current projects include:
Plasma-based Ambient Desorption/Ionization Mass Spectrometry (ADI-MS) for direct surface analysis of organic molecules in complex samples
Direct surface analysis of solid and liquid samples can be performed by ambient desorption/ionization mass spectrometry (ADI-MS) with little to no sample pretreatment and low limits of detection.
We are interested in the optimization and application of plasma-based ambient ionization sources (such as FAPA and LTP) coupled to high resolution mass spectrometry (Orbitrap) for fast screening and identifaction of organic molecules as alternative means to classical analysis protocols that typically involve lenghty chromatographic separations before mass spectrometric detection. In addition, we interested to develop improved quantitation protocols for ambient desorption/ionization mass spectrometry. Applications include lithium-ion battery electrolytes, liquid crystal displays, counterfeit pharmaceuticals, polymers, and other types of samples.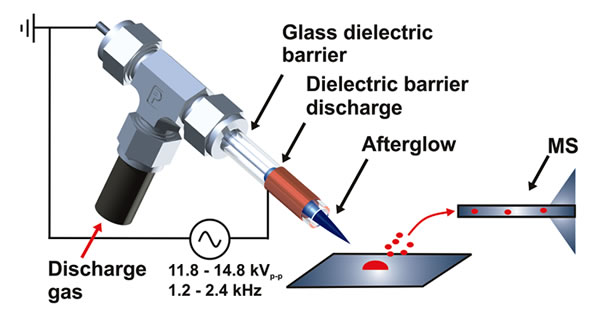
Related Publications:
C. Kuhlmann, J. T. Shelley, C. Engelhard: Plasma-based Ambient Desorption/Ionization Mass Spectrometry for the Analysis of Liquid Crystals Employed in Display Devices,
J. Am. Soc. Mass Spectrom., 2019, 30(10), 2101-2113 (DOI: 10.1007/s13361-019-02280-w).
C. Kuhlmann, M. Heide, C. Engelhard: Fast Screening and Quantitative Mass Spectral Imaging of Thin-Layer Chromatography Plates with Flowing Atmospheric-Pressure Afterglow High-Resolution Mass Spectrometry,
Anal. Bioanal. Chem., 2019, 411(23), 6213-6225 (DOI: 10.1007/s00216-019-02013-8).
D. N. Ateacha, C. Kuhlmann, C. Engelhard: Rapid Screening of Antimalarial Drugs Using Low-Temperature Plasma Desorption/Ionization Orbitrap Mass Spectrometry, Anal. Methods, 2019, 11, 566-574 (DOI: 10.1039/C8AY02538J)
Critical Review: J. T. Shelley, S. P. Badal, C. Engelhard, H. Hayen:
Ambient Desorption/Ionization Mass Spectrometry: Evolution from Rapid Qualitative Screening to Accurate Quantification Tool, Anal. Bioanal. Chem., 2018, 410(17), 4061-4076 (DOI: 10.1007/s00216-018-1023-9
Critical Review: A. Albert et al., Anal. Bioanal. Chem., 2014, 406(25), 6111-6127 (DOI:10.1007/s00216-014-7989-z). Featured by Anal. Bioanal. Chem. on the Cover of issue 25.
Nanoanalysis and Nanomaterials Characterization with ICP-MS in Single Particle Mode and Microsecond Time Resolution
Inductively coupled plasma mass spectrometry (ICP-MS) is a powerful tool for ultra trace analysis. In our group, ICP-MS is used for the detection, identification, and characterization of single nanoparticles in complex matrices. We are interested in pushing the boundaries of ICP-MS instrumentation and have developed a in-house built data aquisition system (DAQ) for ICP-MS that features microsecond time resolution and single particle detection with 100% duty cycle. The µsDAQ provides high-speed, high-quality single-particle ICP-MS results.
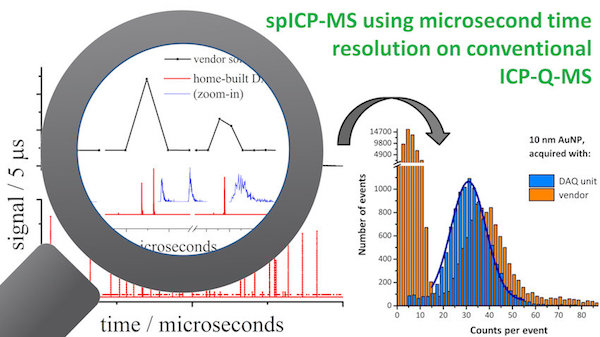
Selected Publications:
I. Strenge, C. Engelhard: Single particle inductively coupled plasma mass spectrometry: investigating nonlinear response observed in pulse counting mode and extending the linear dynamic range by compensating for dead time related count losses on a microsecond timescale,
J. Anal. At. Spectrom., 2020,35, 84-99 (DOI: 10.1039/C9JA00327D).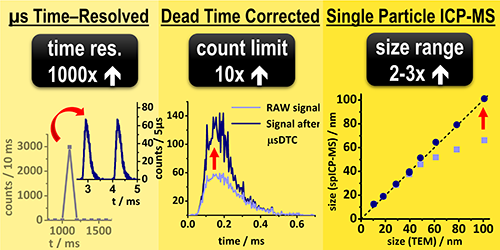
D. Mozhayeva, C. Engelhard: A Quantitative Nanoparticle Extraction Method for Microsecond Time Resolved Single-Particle ICP-MS Data in the Presence of High Background,
J. Anal. At. Spectrom., 2019, 34, 1571-1580 (DOI: doi.org/10.1039/C9JA00042A).
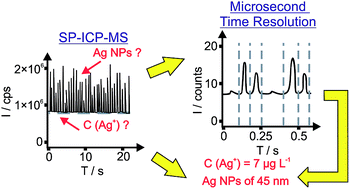
I. Strenge, C. Engelhard: Capabilities of fast data acquisition with microsecond time resolution in inductively coupled plasma mass spectrometry and identification of signal artifacts from millisecond dwell times during detection of single gold nanoparticles,
J. Anal. At. Spectrom., 2016, 31(1), 135-144 (DOI:10.1039/c5ja00177c).
B. Franze, I. Strenge, C. Engelhard: Single Particle Inductively Coupled Plasma Mass Spectrometry: Evaluation of Three Different Pneumatic and Piezo-based Sample Introduction Systems for the Characterization of Silver Nanoparticles, et al., J. Anal. At. Spectrom., 2012, 27(7), 1074-1083 (DOI: 10.1039/C2JA00003B).
Capillary electrophoresis coupled to elemental mass spectrometry (CE-ICP-MS, CE-spICP-MS) for nanomaterials fractionation and characterization
In this project, capillary electrophoresis (CE) is coupled to single particle inductively coupled plasma mass-spectrometry (SP-ICP-MS) and a prototype data acquisition (μsDAQ) system to separate and quantify mixtures of different types of nanoparticles. We demonstrated the fractionation and quantification of gold and silver nanoparticles with CE-ICP-MS in the past and are currently expanding our efforts to nanoparticles with different surface chemistries. Also, we are developing sample pre-concentration methods to be able to analyze nanoparticles in environmental samples with CE-SP-ICP-MS.
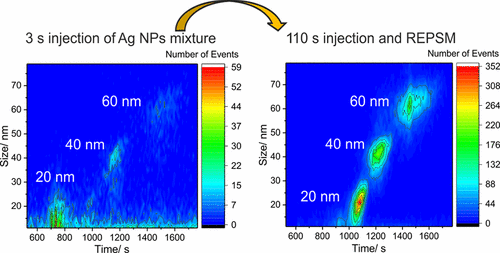
Related Publications:
D. Mozhayeva, C. Engelhard*:
Separation of Silver Nanoparticles with Different Coatings by Capillary Electrophoresis Coupled to ICP-MS in Single Particle Mode,
Anal. Chem., 2017, 89, 9767-9774 (DOI: 10.1021/acs.analchem.7b01626).
D. Mozhayeva, I. Strenge, C. Engelhard*:
Implementation of Online Pre-Concentration and Microsecond Time Resolution to Capillary Electrophoresis Single Particle Inductively Coupled Plasma Mass Spectrometry (CE-SP-ICP-MS) and its Application in Silver Nanoparticle Analysis,
Anal. Chem., 2017, 89, 7152-7159 (DOI: 10.1021/acs.analchem.7b01185).
B. Franze, I. Strenge, C. Engelhard*:
Separation and Detection of Gold Nanoparticles with Capillary Electrophoresis and ICP-MS in Single Particle Mode (CE-SP-ICP-MS),
J. Anal. At. Spectrom., 2017, 32, 1481-1489 (DOI: 10.1039/C7JA00040E).
Imaging Mass Spectrometry and Surface Analysis by ToF-SIMS
Time-of-Flight Secondary Ion Mass Spectrometry (ToF-SIMS) is an excellent tool for qualitative elemental and molecular surface analysis, depth profiling, and imaging mass spectrometry. We have a ToF-SIMS IV (IONTOF) in our laboratory and use it for fast surface analysis and depth profiling of layered materials. In a collaboration with Tascon GmbH, we are also interested to push the boundaries of this technique and to develop methods for high-resolution, high-sensitivity nanoparticle detection in complex matrices.
The instrument is part of the Micro- and Nanoanalytics Facility of the University of Siegen and can be used by other trained users across the University and beyond. For details on this instrument and other available core facility instrumentation, please visit the Equipment page of the Micro- and Nanoanalytics Facility.

Image: André Silz, USI
Related Publications:
D. N. Ateacha, U. Koch, C. Engelhard*:
Direct Analysis of Alkaloids in Natural Cinchona Bark and Commercial Extracts using Time-of-Flight Secondary Ion Mass Spectrometry,
Anal. Methods, 2018, published online (DOI: 10.1039/C7AY02822A).
L. Veith*, J. Böttner, A. Vennemann, D. Breitenstein, C. Engelhard, J. Meijer, I. Estrela-Lopis, M. Wiemann*, B. Hagenhoff:
Detection of ZrO2 Nanoparticles in Lung Tissue Sections by Time-of-Flight Secondary Ion Mass Spectrometry and Ion Beam Microscopy,
Nanomaterials, 2018, 8(1), 44 (DOI: 10.3390/nano8010044).
L. Veith*, D. Dietrich, A. Vennemann, D. Breitenstein, C. Engelhard, U. Karst, M. Sperling, M. Wiemann, B. Hagenhoff:
Combination of Micro X-Ray Fluorescencee Spectroscopy and Time-of-Flight Secondary Ion Mass Spectrometry Imaging for the Marker-free Detection of NM212 CeO2 Nanoparticles in Tissue Sections,
J. Anal. At. Spectrom., 2018, published online (DOI: 10.1039/C7JA00325K)

