Research
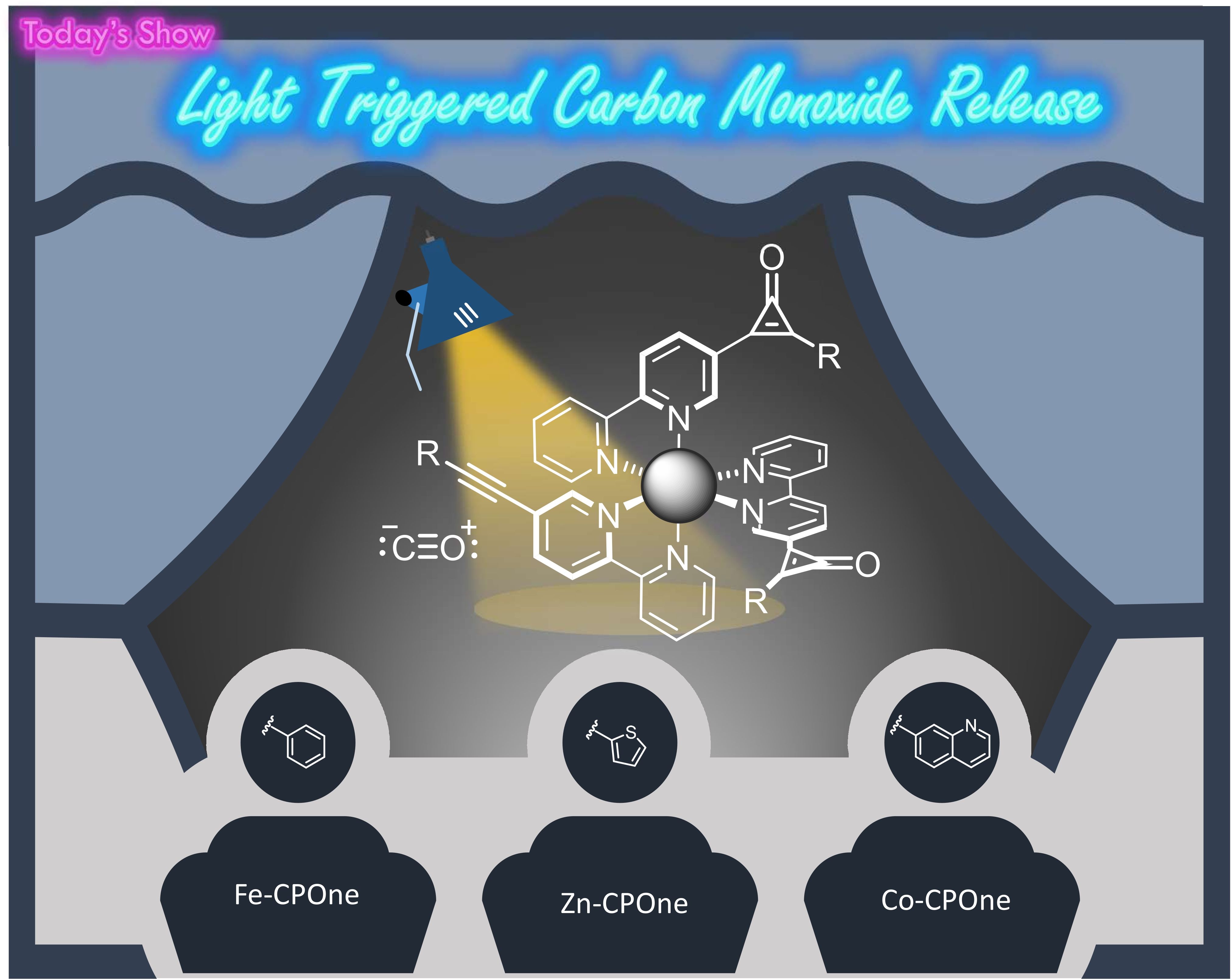
Cyclopropenones are the smallest aromatic system and despite containing a strained three-membered ring, they are surprisingly stable due to this aromaticity. Upon irradiation cyclopropenones are known to release carbon monoxide in a process known as photodecarbonylation, resulting in the corresponding alkyne.
Our group has reported M-CPOnes, transition metal complexes with cyclopropenone-containing ligands, for light-triggered CO release. The cyclopropenone is functionalised with electron-deficient N-heterocycles for metal coordination and the wavelength for photodecarbonylation can be tuned by introducing different transition metals and cyclopropenone substituents (Dalton Trans., 2022, 51, 6936 - 6943).
Spin-Crossover FeII Complexes and Metal-Organic Cages
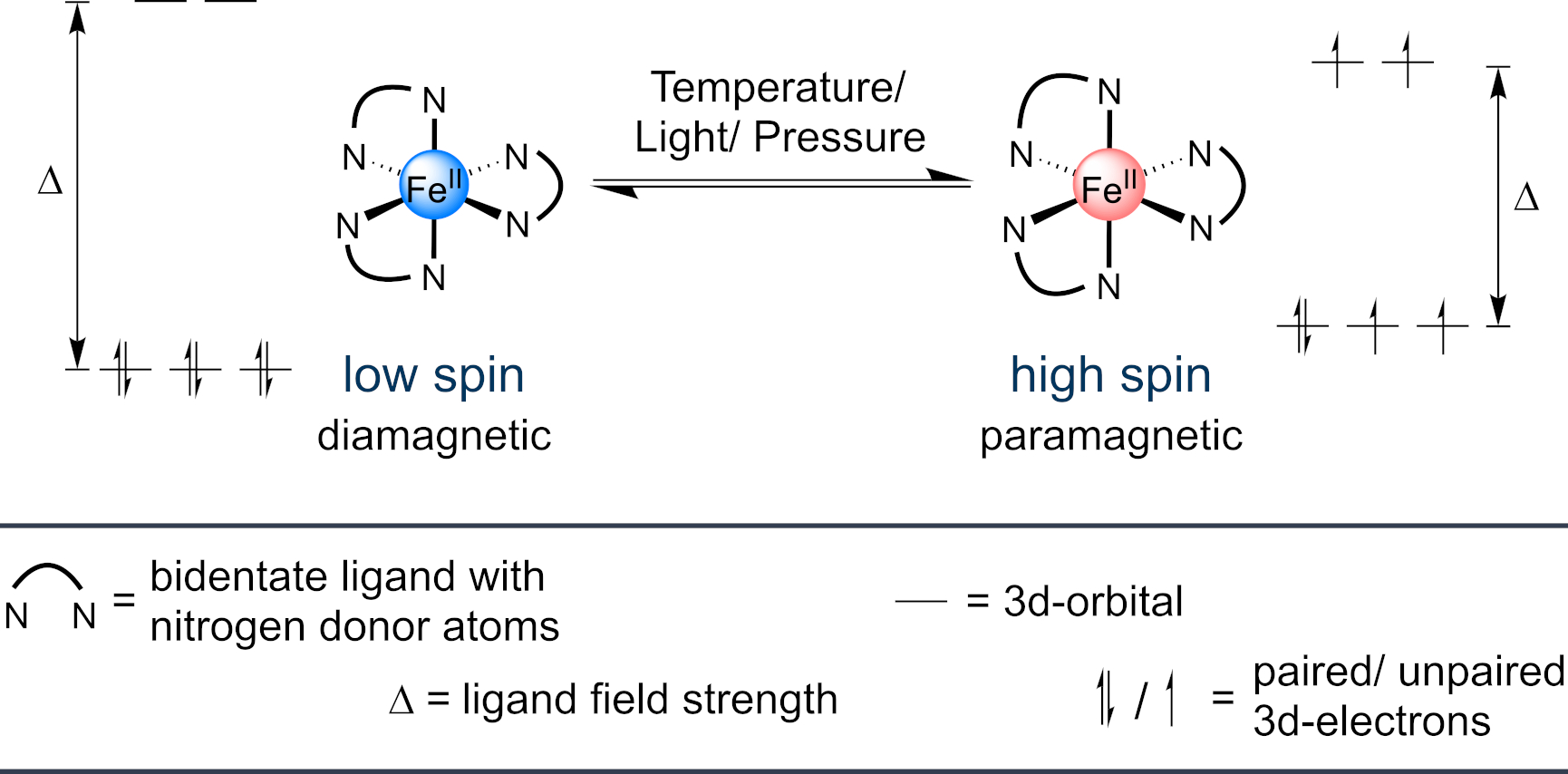
Octahedrally coordinated transition metal ions with 3d4 to 3d7 electronic configurations can occur in two spin states if their ligand field strength (Δ) and spin-pairing energy are of a similar magnitude. A greater ligand field strength compared to spin pairing energy results in the adoption of the diamagnetic low spin state, with the maximum of paired electrons. Vice versa, a greater spin pairing energy compared to ligand field strength leads to the paramagnetic high spin state, with the maximum of unpaired electrons. The transition of the two states is termed spin-crossover. Interestingly, the ligand field strength can be manipulated by external stimuli like temperature, light and pressure, which makes spin-crossover complexes attractive for magnetic switching or sensing applications.
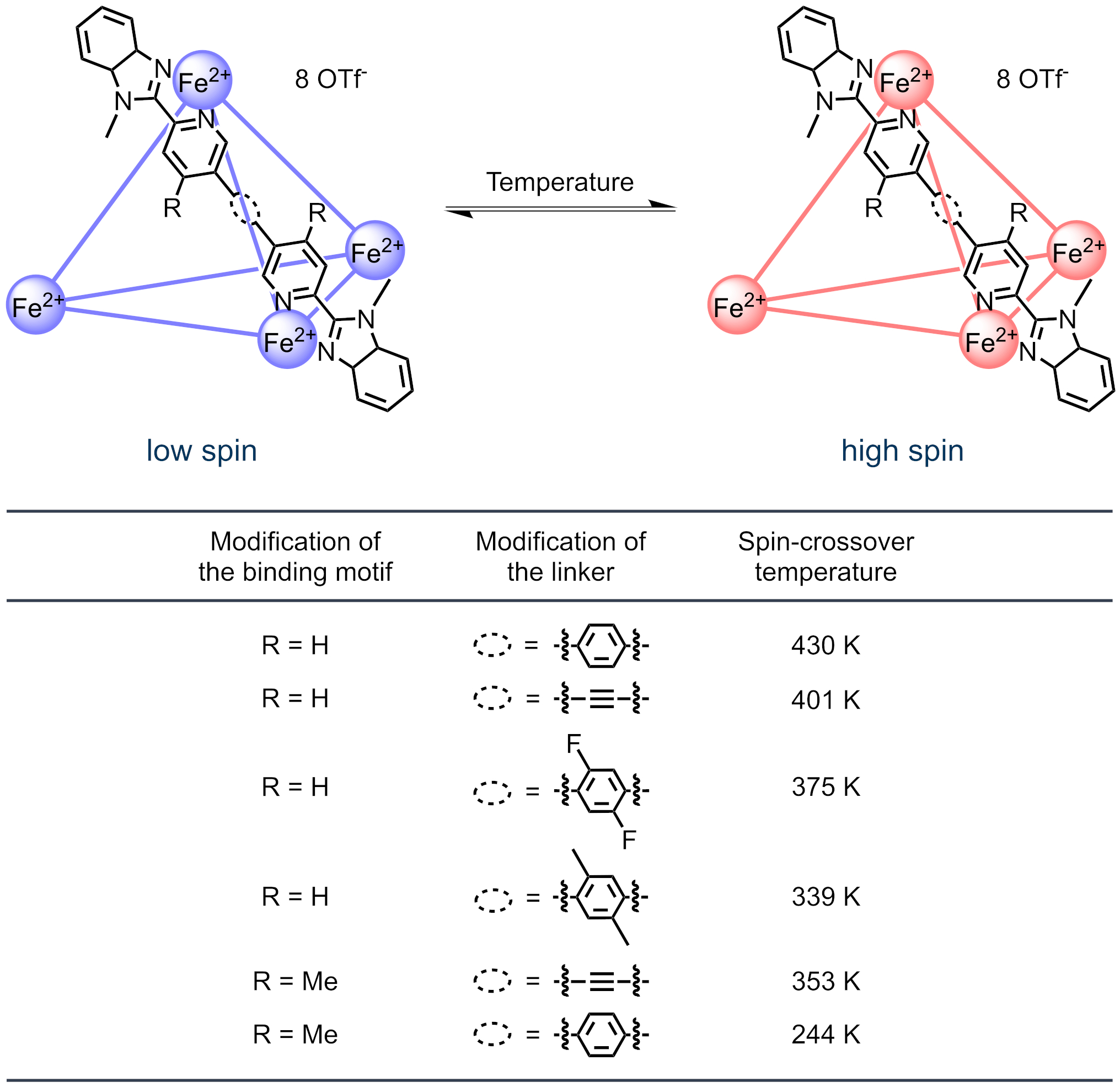
Our group is interested in the design of polynuclear spin-crossover complexes or, to be more precise, spin-crossover cages. We have recently shown that tuning of the spin-crossover temperature (temperature with equal spin state fractions) towards room temperature is possible in tetrahedral FeII4L6 cages with linear 2-(2’-pyridyl)benzimidazole ligands by introduction of relatively subtle ligand modifications (Dalton Trans., 2023, 52, 12789 - 12795).
The Synthesis and Dynamic Covalent Chemistry of Amidoboronates
BN containing heterocycles can give access to different and interesting reactivity as well as functionality compared to their carbon analogues. Amidoboronates are a novel class of BN containing heterocycles readily synthesised from N-aryl iminoboronates through a reductive coupling using a single electron reductant ( Dalton Trans., 2023 , 52, 9189 - 9201).
The reductive coupling yields up to 3 different amidoboronate isomers, the diastereomeric rac5 and meso5 products and the fused six-membered rac6 ring system (
Chem. Eur. J.,
2018
,
24
, 1200 - 12005
). Amidoboronates potentially contain up to 3 types of dynamic covalent bonds including previously unknown dynamic B-N bonds. We have shown that the rac5/rac6 isomeric ratio can be influenced through variation of the para-aniline substituent and catechol sub-components through exploitation of the dynamic covalent B-N and B-O bonds (
ChemPlusChem
,
2022
,
87
, e20220022
).
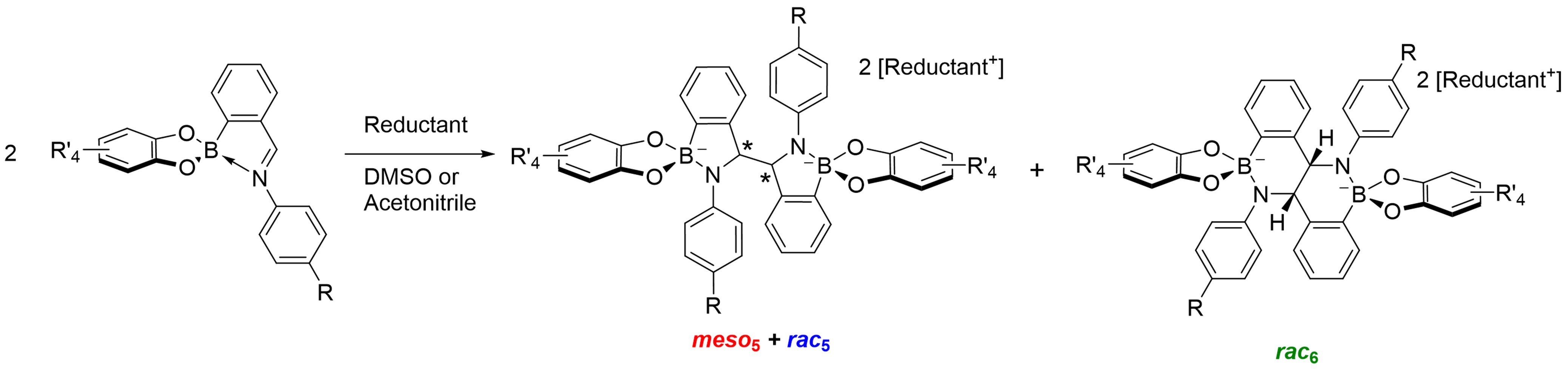
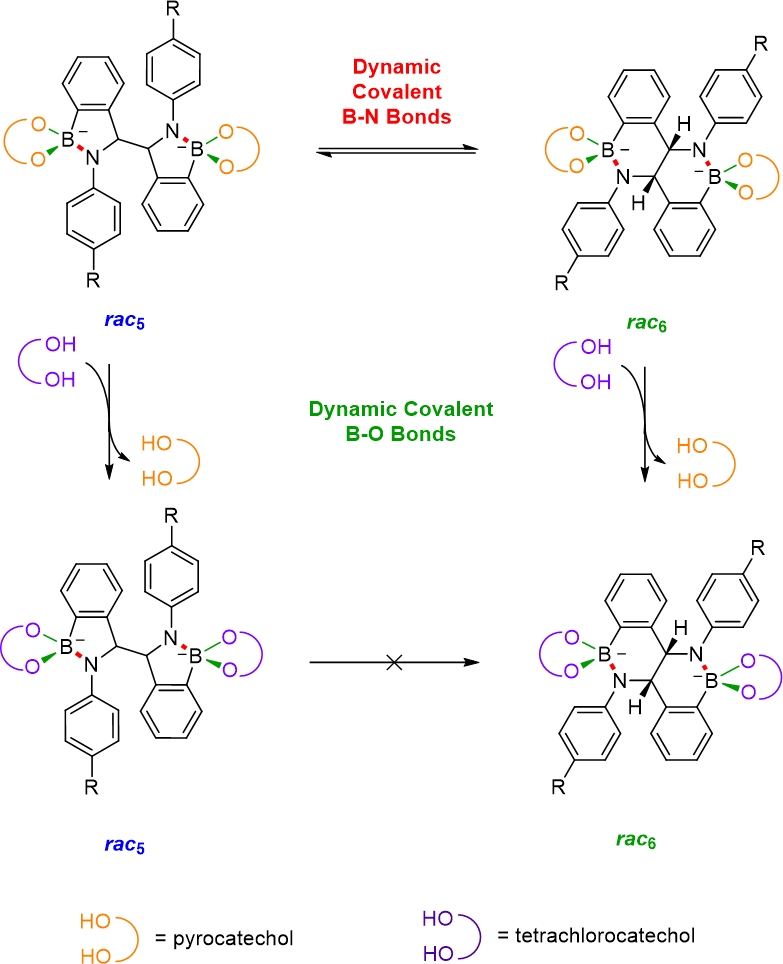
Figure from ChemPlusChem 2022
, 87, e20220022
BN containing heterocycles can give access to different and interesting reactivity as well as functionality compared to their carbon analogues. Amidoboronates are a novel class of BN containing heterocycles readily synthesised from N-aryl iminoboronates through a reductive coupling using a single electron reductant ( Dalton Trans., 2023 , 52, 9189 - 9201).
The reductive coupling yields up to 3 different amidoboronate isomers, the diastereomeric rac5 and meso5 products and the fused six-membered rac6 ring system ( Chem. Eur. J., 2018 , 24 , 1200 - 12005 ). Amidoboronates potentially contain up to 3 types of dynamic covalent bonds including previously unknown dynamic B-N bonds. We have shown that the rac5/rac6 isomeric ratio can be influenced through variation of the para-aniline substituent and catechol sub-components through exploitation of the dynamic covalent B-N and B-O bonds ( ChemPlusChem , 2022 , 87 , e20220022 ).


Figure from ChemPlusChem 2022 , 87, e20220022
